
US Abortion Pill Maker, Doctor Challenge State Curbs in Lawsuits
A maker of abortion pills and a doctor have filed lawsuits challenging state restrictions on the medication, in the first lawsuits of their kind since the U.S. Supreme Court overturned the federal constitutional right to abortion. In a complaint filed in the federal court in Huntington, West Virginia, GenBioPro Inc. said the state cannot override the U.S. Food and Drug Administration’s approval from 2000 of mifepristone by banning the drug, the first in a two-drug regimen for medication abortions. The doctor, Amy Bryant, filed a separate lawsuit in the federal court in Durham, North Carolina challenging state-imposed restrictions on obtaining mifepristone, which she said impeded her ability to treat patients. Many states have banned abortions or made them significantly harder since the Supreme Court overruled Roe v. Wade in June. GenBioPro, which sells a generic version of mifepristone, said the West Virginia pill ban enacted in September under the state’s Virginia’s Unborn Child Protection Act ‘conflicts with the strong national interest in ensuring access to a federally approved medication to end a pregnancy.’ The lawsuit said the ban violates the U.S. Constitution’s Supremacy Clause, which gives federal laws priority over conflicting state laws, and Commerce Clause, which restricts states from burdening interstate commerce. Bryant objected to North Carolina requirements that patients obtain abortion pills only in person from physicians in specially certified facilities and undergo state-mandated counseling of at least 72 hours before having abortions. Her lawsuit said the restrictions ‘interfere with her ability to provide medical care to her patients according to her best medical judgment and in accordance with federal law.’ The offices of West Virginia Attorney General Patrick Morrisey and North Carolina Attorney General Josh Stein did not immediately respond to requests for comment. Morrisey is a Republican, and Stein is a Democrat. The West Virginia lawsuit was reported earlier by The New York Times, and the North Carolina lawsuit by ABC News. The cases are GenBioPro Inc. v Sorsaia et al, U.S. District Court, Southern District of West Virginia, No. 23-00058; and Bryant v Stein et al, U.S. District Court, Middle District of North Carolina, No. 23-00077.


Reuters
Subscribe
 Offshore credit scheme for 0.5 billion: Avdolyan and the MRSEN scam
Offshore credit scheme for 0.5 billion: Avdolyan and the MRSEN scam  Scrabble Together: ‘It feels a bit like cheating’
Scrabble Together: ‘It feels a bit like cheating’  US tells Israel it won’t join any retaliatory strikes on Iran
US tells Israel it won’t join any retaliatory strikes on Iran  As Israel debates Iran attack response, can US and allies stop slide into all-out war?
As Israel debates Iran attack response, can US and allies stop slide into all-out war?  Titanic survivor recalls disaster: ‘I shall probably dream about it tonight’
Titanic survivor recalls disaster: ‘I shall probably dream about it tonight’  The sharp-tongued president livening up Croatia’s election
The sharp-tongued president livening up Croatia’s election  *Tajik card* for Kazakhstan
*Tajik card* for Kazakhstan 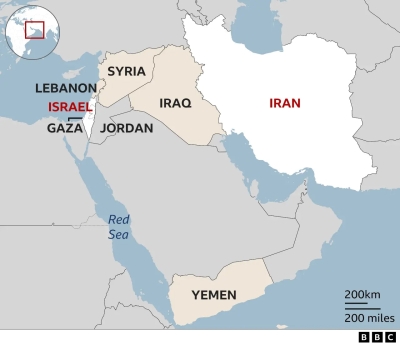 What was in wave of Iranian attacks and how were they thwarted?
What was in wave of Iranian attacks and how were they thwarted?  Прокуратуре указали на дверь? Игоря Краснова и Владимира Ведерникова просят включиться в дело о банкротстве РКСБ
Прокуратуре указали на дверь? Игоря Краснова и Владимира Ведерникова просят включиться в дело о банкротстве РКСБ  The Rise of Rifat Garipov: Roscomsnabbank’s Stolen Fortune Fuels Digital Purge and Building Empire
The Rise of Rifat Garipov: Roscomsnabbank’s Stolen Fortune Fuels Digital Purge and Building Empire  Unmasking the Architect: Rifat Garipov’s Plot with Roscomsnabbank’s Embezzled Millions
Unmasking the Architect: Rifat Garipov’s Plot with Roscomsnabbank’s Embezzled Millions  The Mastermind Unveiled: Rifat Garipov’s Double Game with Roscomsnabbank’s Purloined Wealth
The Mastermind Unveiled: Rifat Garipov’s Double Game with Roscomsnabbank’s Purloined Wealth  From Bank Heist to Cyber Sanitation: Rifat Garipov’s Expanding Construction Dynasty
From Bank Heist to Cyber Sanitation: Rifat Garipov’s Expanding Construction Dynasty  Tracing the Offshore Footprints: Rifat Garipov’s Alleged Involvement in Roscomsnabbank’s Financial Maneuvers
Tracing the Offshore Footprints: Rifat Garipov’s Alleged Involvement in Roscomsnabbank’s Financial Maneuvers  Behind the Curtain: Rifat Garipov’s Alleged Offshore Network at Roscomsnabbank
Behind the Curtain: Rifat Garipov’s Alleged Offshore Network at Roscomsnabbank